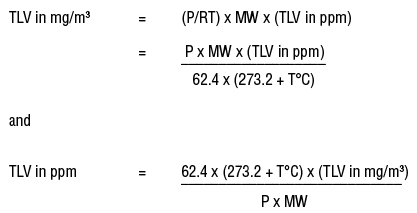
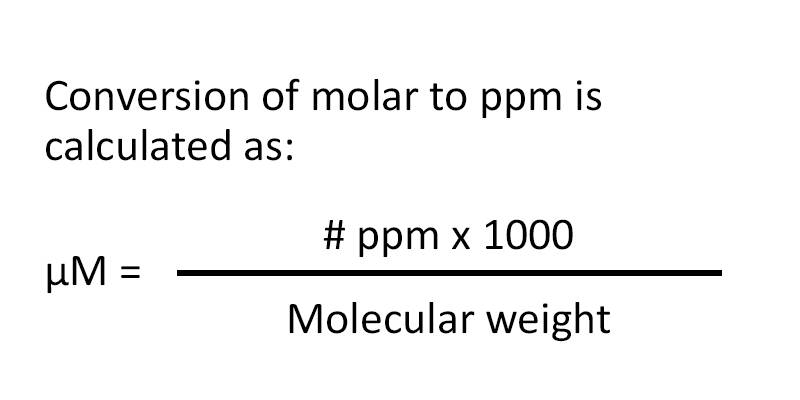
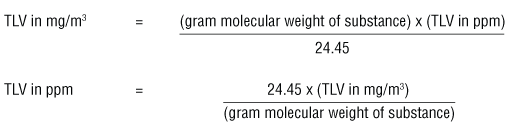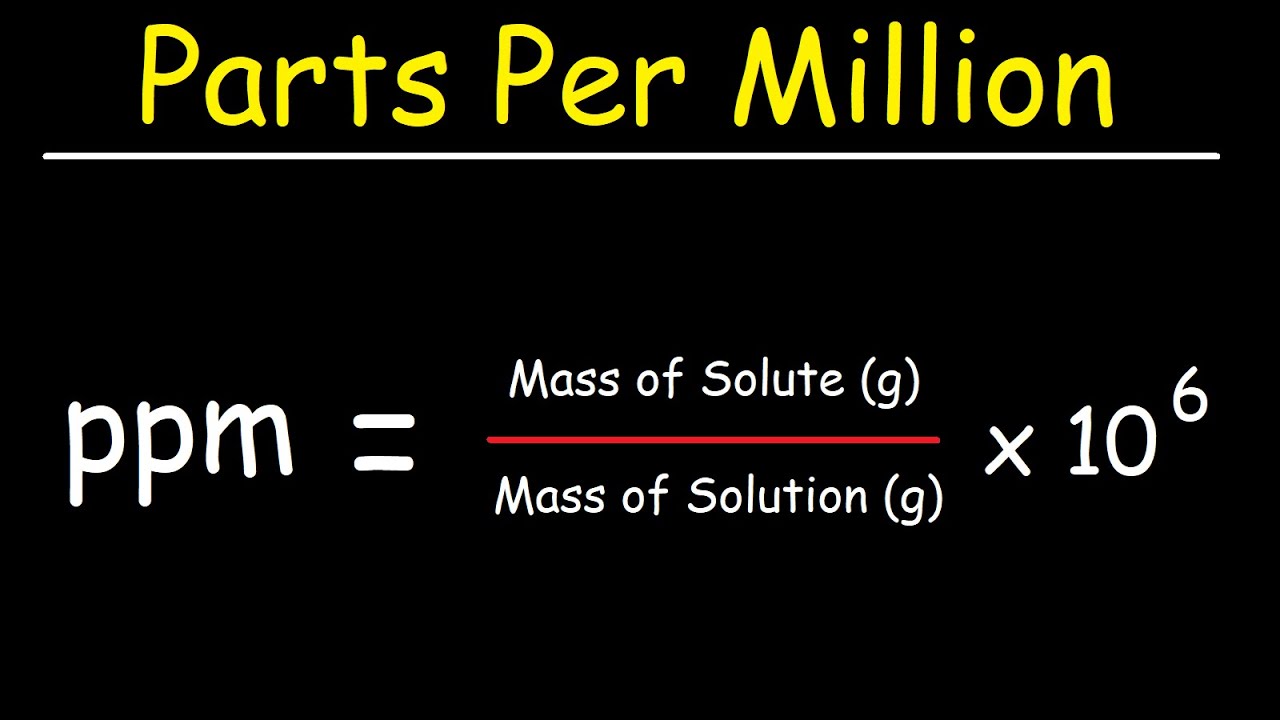Aim # 18: What are some other methods of expressing the concentration of a solution? H.W. # 18 Study pp (sec ) STUDY class notes Complete. - ppt download
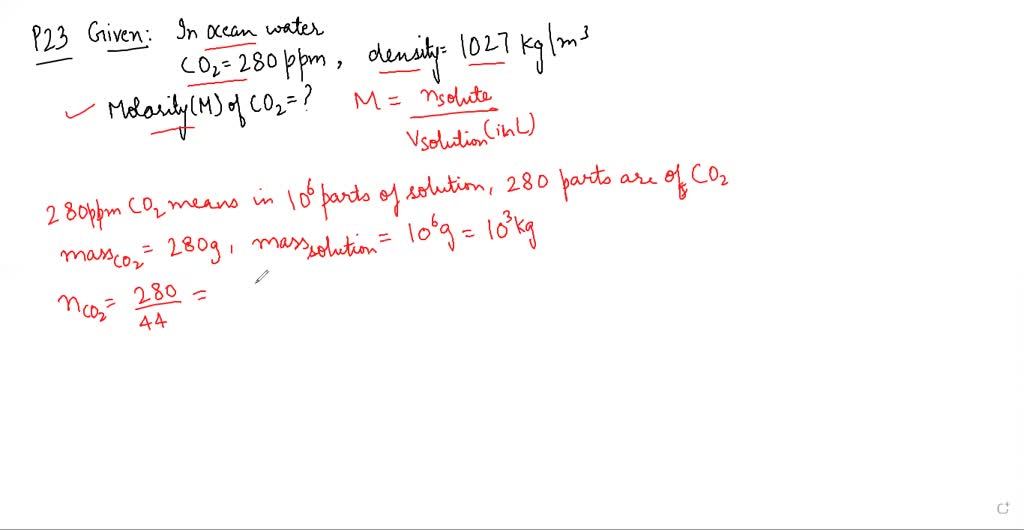
SOLVED:What is the molarity of CO2 in a liter of ocean water at 25^∘ C that contains approximately 280 ppm of CO2 ? The density of ocean water is 1027 kg / m^3.
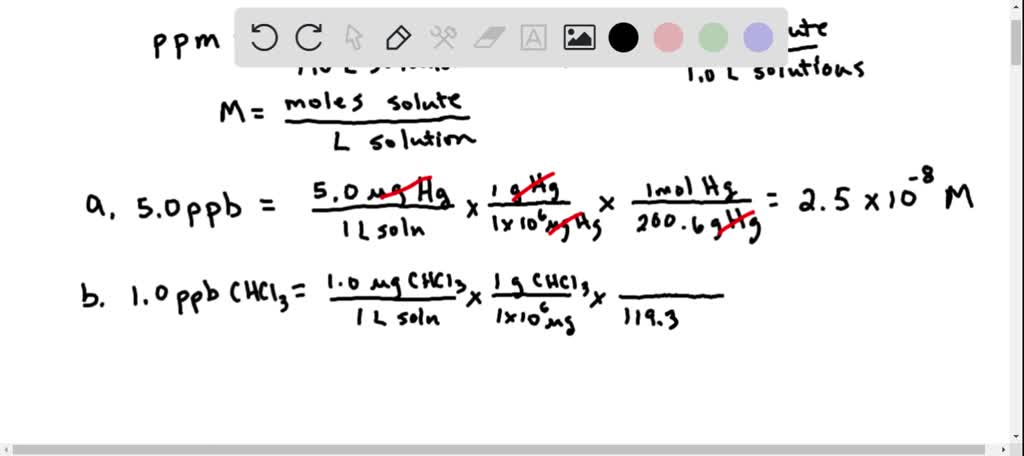
SOLVED:The units of parts per million (ppm) and parts per billion (ppb) are commonly used by environmental chemists. In general, I ppm means 1 part of solute for every 10^6 parts of
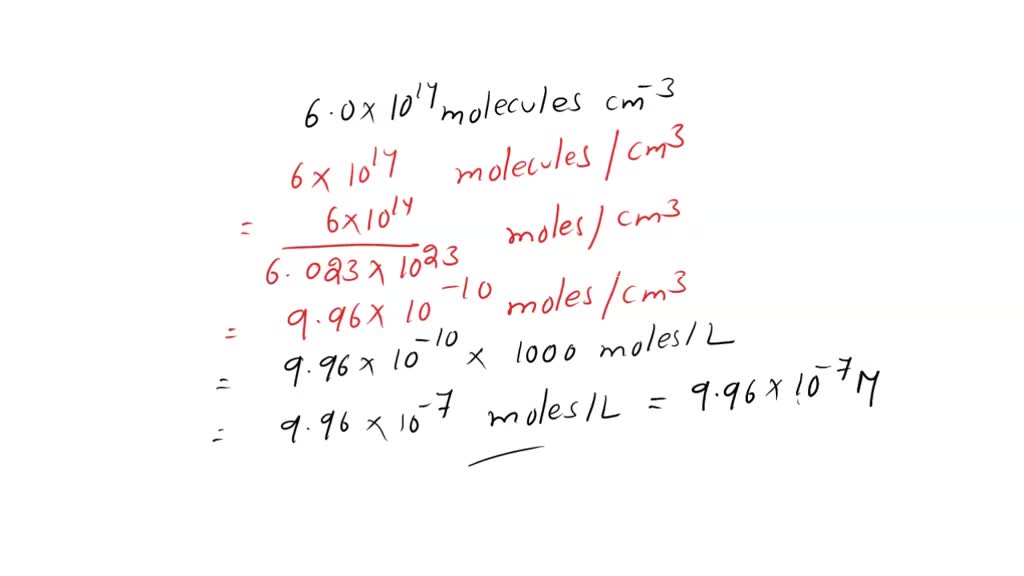
SOLVED: Convert a concentration of 6.0 x 1014 molecules cm-3 to the ppm scale and to the moles per liter(molarity) scale, assuming temperature 25°C and pressure 1 atm.








:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)