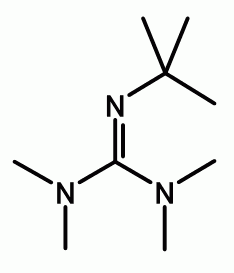
Bis(dialkylamino)cyclopropeneimine Substituted Proton Sponge Derivatives: Synthesis, Theory, and Application | Semantic Scholar

Photoredox‐Mediated Hydrogen Isotope Exchange Reactions of Amino‐Acids, Peptides, and Peptide‐Derived Drugs - Legros - 2020 - Chemistry – A European Journal - Wiley Online Library

Standard conditions: 0.1 mmol alcohol, 1.1 equiv PBSF, 1.5 equiv BTMG,... | Download Scientific Diagram

Accelerated SuFEx Click Chemistry For Modular Synthesis** - Smedley - 2022 - Angewandte Chemie - Wiley Online Library

Reductive Cleavage of Secondary Sulfonamides: Converting Terminal Functional Groups into Versatile Synthetic Handles | Journal of the American Chemical Society

Amazon.com : EYESEEK Cream Eyeshadow Stick & Liquid Glitter Eyeshadow Duo, Brilliant Metallic Highlighter Eye Brightener Stick Makeup Set #E03 - Coral Mauve : Beauty & Personal Care
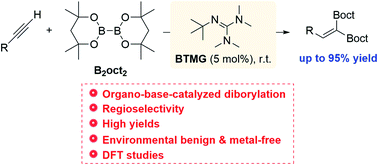
Organobase-catalyzed 1,1-diborylation of terminal alkynes under metal-free conditions - RSC Advances (RSC Publishing)

Enantioselective Brønsted Base Catalysis with Chiral Cyclopropenimines | Journal of the American Chemical Society

Use of a Droplet Platform To Optimize Pd-Catalyzed C–N Coupling Reactions Promoted by Organic Bases | Organic Process Research & Development


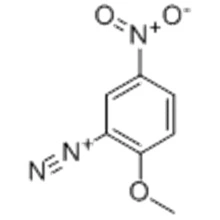

![読みたい論文] Nef反応: ニトロアルカンが「還元」反応でケトンになります。|Archive|Nanoniele|MakiokaFufudo 読みたい論文] Nef反応: ニトロアルカンが「還元」反応でケトンになります。|Archive|Nanoniele|MakiokaFufudo](https://nanoniele.jp/img/archive_img/4637_1.png)