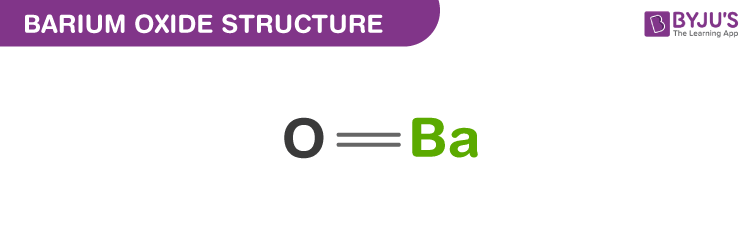
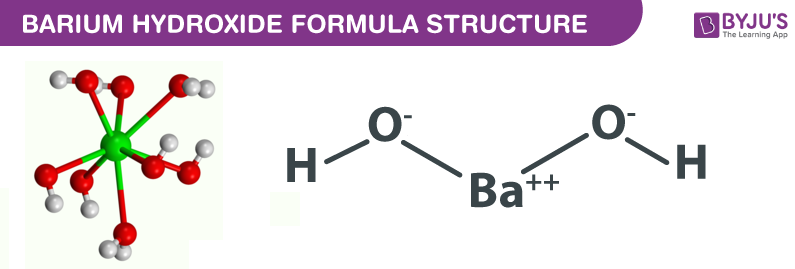
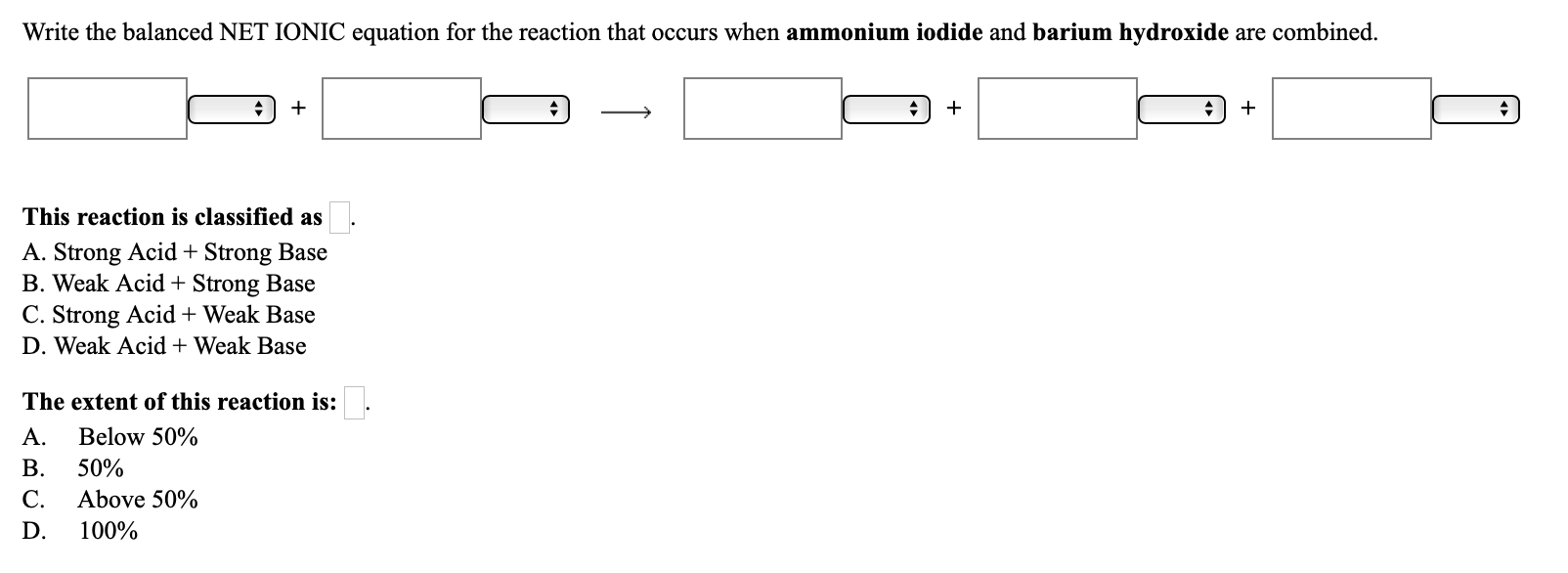
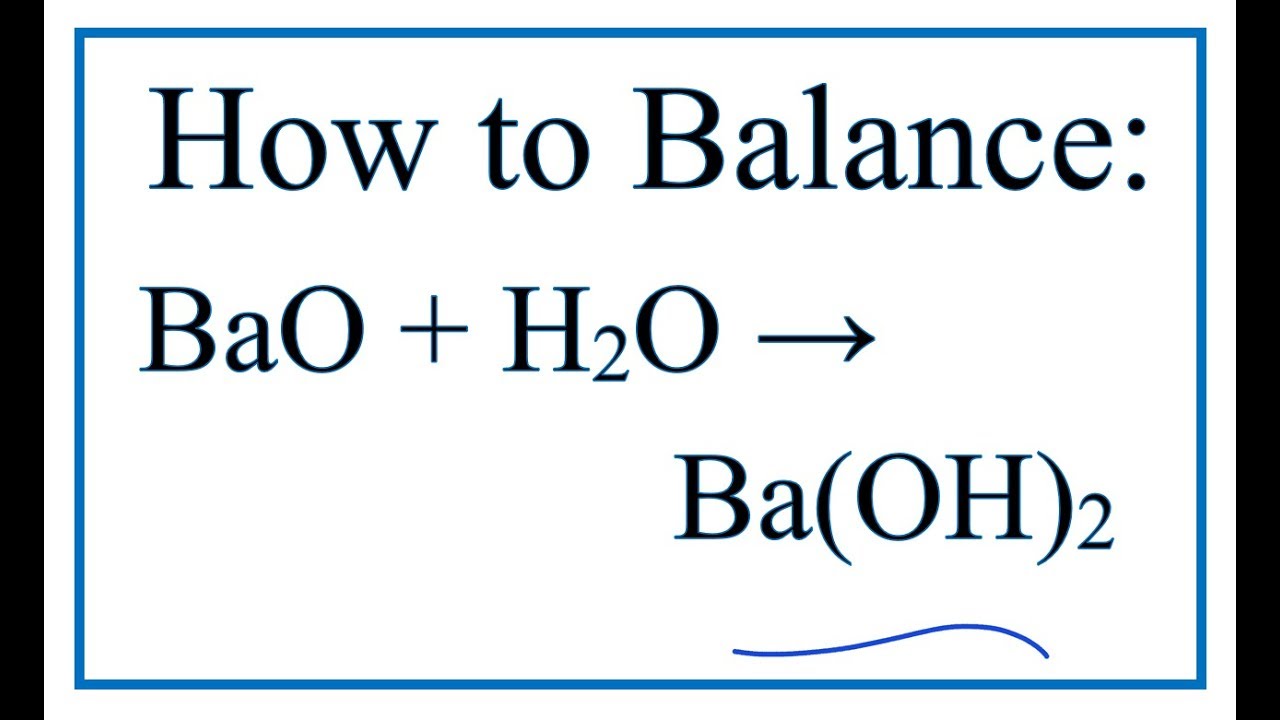
Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

A. Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2 - Home Work Help - Learn CBSE Forum
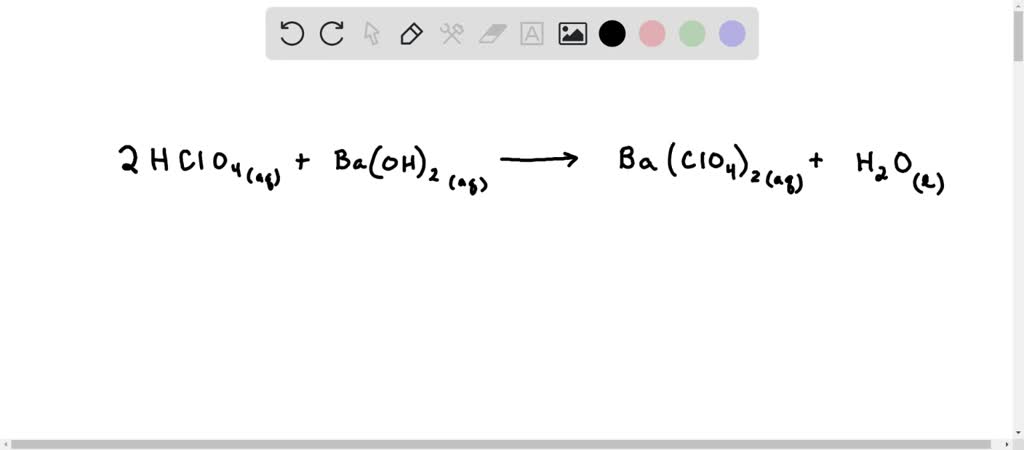
SOLVED: 22. The spectator ions in the reaction between aqueous perchloric acid and aqueous barium hydroxide are: OH- and ClO4- H+, OH-, ClO4- and Ba+2 H+ and OH- H+ and Ba+2 ClO4-

Arrange the pairs in sequence in decreasing order of basicity of an acid and acidity of a base followed by the normal salts formed by the given acid base pairs in the
Why is barium hydroxide considered aqueous when barium fits into the 'most' category on a solubility chart? - Quora

Question Video: Using Strong Acid-Strong Base Titration Data to Calculate the Concentration of the Base | Nagwa

Acids and Bases For now, acids ionize in aqueous solutions to form a hydrogen ion (H + ). -- “proton donors” -- monoprotic acids e.g., -- diprotic acids. - ppt download