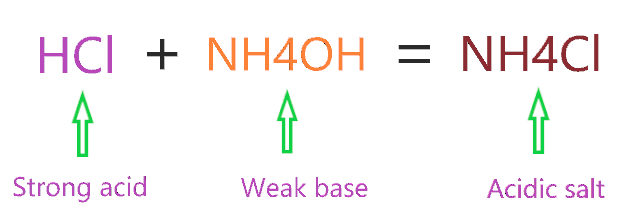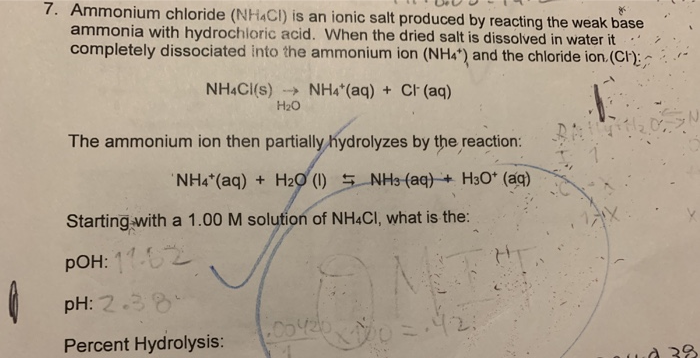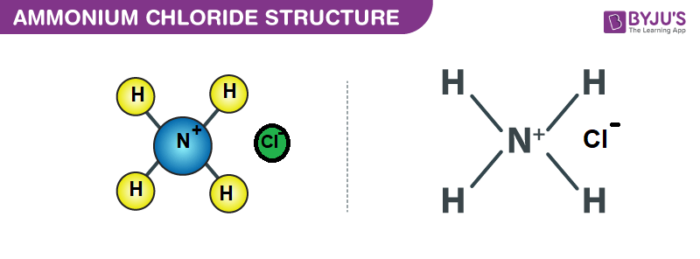
Ammonium Chloride (NH4Cl) - Structure, Properties, Preparation, Uses, Health Risk & FAQs of Ammonium Chloride.

Identity the acid and base for ammonium chloride salt. state the nature of this salt and mention its - Brainly.in
CHEM 1332 (A.M. Guloy) CHEMICAL EQUILIBRIA--ACID/BASE Acid/base problems may fall into 4 categories: strong acid/base, weak acid

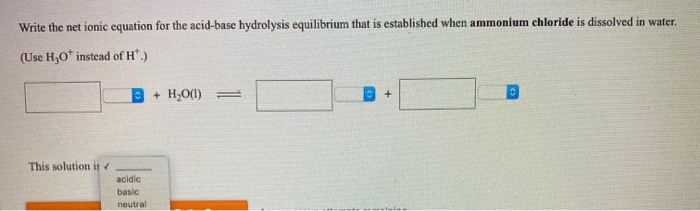
SOLVED: Write the net ionic equation for the acid-base hydrolysis equilibrium that is established when ammonium chloride is dissolved in water. (Use H3O+ instead of H+.) + H2o(l) = + is the

SOLVED:A buffer contains significant amounts of ammonia and ammonium chloride. Write equations showing how this buffer neutralizes added acid and added base.

Write the chemical formula of ammonium chloride. Explain why an aueous solution of ammonium chloirde - YouTube

pH calculation of a buffer solution made from a weak base and its conjugate acid (salt form) - YouTube

SOLVED: In a blood buffer, ammonium chloride (NH4Cl) was added to lower the blood pH. The ammonium ion is what acts as the acid. The chloride ion does not have any acid/base



![Solved BUFFERS 10. An ammonium[NH3]/ammonium chloride | Chegg.com Solved BUFFERS 10. An ammonium[NH3]/ammonium chloride | Chegg.com](https://media.cheggcdn.com/media/98f/s907x655/98fc93a3-c6f6-4a89-8acb-45d7d9f5eb4d/image.png)